

West Jordan, Utah — April 13, 2026
Route 92 Medical, a U.S.-based medtech company focused on neurovascular intervention, has raised $50 million in growth financing to expand its commercial footprint and advance its stroke treatment platform.
The funding round was led by Novo Holdings, with participation from new investor Sectoral Asset Management and existing backers including U.S. Venture Partners, Norwest Venture Partners, InnovaHealth Partners, and The Vertical Group.
The company plans to use the capital to accelerate global commercialization, strengthen clinical evidence supporting its technology, and continue investing in product innovation.
Route 92 Medical is a medtech company founded in 2015. The company develops devices for treating acute ischemic stroke, including its HiPoint Reperfusion System, which uses a “Monopoint” approach designed to improve the efficiency of clot removal procedures.
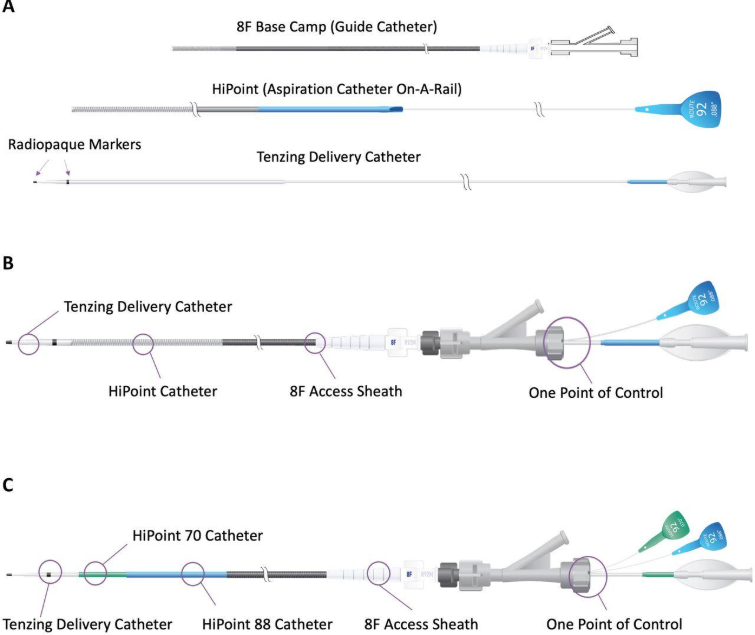
The company is increasingly becoming a Utah-centered operation. While originally based in San Mateo, California, the company has expanded aggressively into the Salt Lake region, establishing a 40,000-square-foot manufacturing and training facility and, more recently, relocating its headquarters to West Jordan.
West Jordan now serves as the company’s primary base, where it plans to create more than 100 high-paying jobs and scale production of its neurovascular devices. Its core products—including the FreeClimb, Tenzing, and HiPoint catheter systems—are designed to work together as an integrated platform that simplifies and accelerates clot removal procedures.
With more than $200 million in funding and growing clinical adoption, Route 92 is transitioning from a development-stage startup into a commercial medtech player, with Utah playing a central role in its manufacturing, talent pipeline, and long-term growth strategy.
In November 2023, the company announced it had raised a $31 million Series F round to accelerate the commercial adoption of a neurovascular intervention portfolio focused on acute ischemic stroke. That capital raise was led by existing venture capital investors: U.S. Venture Partners, Norwest Venture Partners, InnovaHealth Partners, and The Vertical Group.
Investors are backing Route 92 Medical on a simple premise: in stroke care, speed is everything, and current tools are often too slow and inefficient. The company’s integrated system is designed to streamline procedures, navigate complex brain anatomy more effectively, and remove clots faster and more completely. If it delivers on those claims, it could materially improve patient outcomes—making it a high-upside bet in a large and growing market.

CEO and founder Dr. Tony Chou pointed to strong recent adoption of the company’s products, citing clinical performance and growing commercial capabilities as key drivers. Investors echoed confidence in the company’s positioning, highlighting its differentiated technology, manufacturing capacity, and long-term growth potential.
Route 92 Medical seeks to scale adoption of its neurovascular solutions across more hospitals worldwide, targeting improved outcomes for patients suffering from stroke.
Learn more at route92medical.com.

