

Orem, Utah — April 15, 2026
A Long Shot Worth Taking
Tuberculosis has been killing people for millennia. It killed Chopin, Keats, and Orwell. It still kills more than a million people a year, more than almost any other infectious disease on earth. And yet, in a modest lab on the Utah Valley University campus, a professor and a rotating cast of students are quietly working on something that could, eventually, change how the disease is fought.
It's a long shot. Nathan Goldfarb, Associate Professor of Biochemistry at UVU, will tell you that himself.
"This is proof-of-concept work," he said. "We're not saying we have a drug. We're saying we have something worth chasing."

The Problem with the Current Fix
TB is both preventable and treatable, which makes its global death toll all the more frustrating. The drugs exist. The problem is what they ask of the people who take them.
Standard therapy can stretch to six months or more, sometimes longer for drug-resistant strains. The side effects are significant. And patient compliance, which involves a grueling drug regimen for half a year, is one of the central challenges in controlling the disease worldwide.
Part of the reason treatment is so demanding has to do with biology. Mycobacterium tuberculosis, the bacterium that causes TB, is encased in a notoriously thick, waxy cell wall. Getting drugs past that wall requires high doses. High doses mean more toxicity. More toxicity means more patients who stop taking their medication before the infection is fully cleared, which, in turn, accelerates the development of resistance.
It's a feedback loop that has plagued TB treatment for decades.
Disarming Instead of Killing
Goldfarb's lab is trying something different.
His team is going after the bacterium directly, since Hip1 “lives” on the outside of the cell membrane of Mycobacterium tuberculosis. What is different is that the compounds they are using don’t directly kill the bacteria, but weaken it so it can be cleared by the immune system more easily.
The distinction matters.
Most TB drugs are designed to kill the bacterium outright, which requires penetrating that formidable cell wall. Hip1, however, is located on the outside of the organism, which means inhibiting it doesn't require the same kind of chemical brute force.
"This work focuses on disarming the bacteria rather than killing it outright," Goldfarb explained in a conversation with TechBuzz. "By targeting a virulence factor that helps TB evade the immune system, we may be able to improve treatment outcomes while reducing the risk of antibiotic resistance."
In practical terms, that could eventually translate to lower doses, fewer side effects, and treatment courses that are easier for patients to complete. For now, those remain goals, but the early results are encouraging.
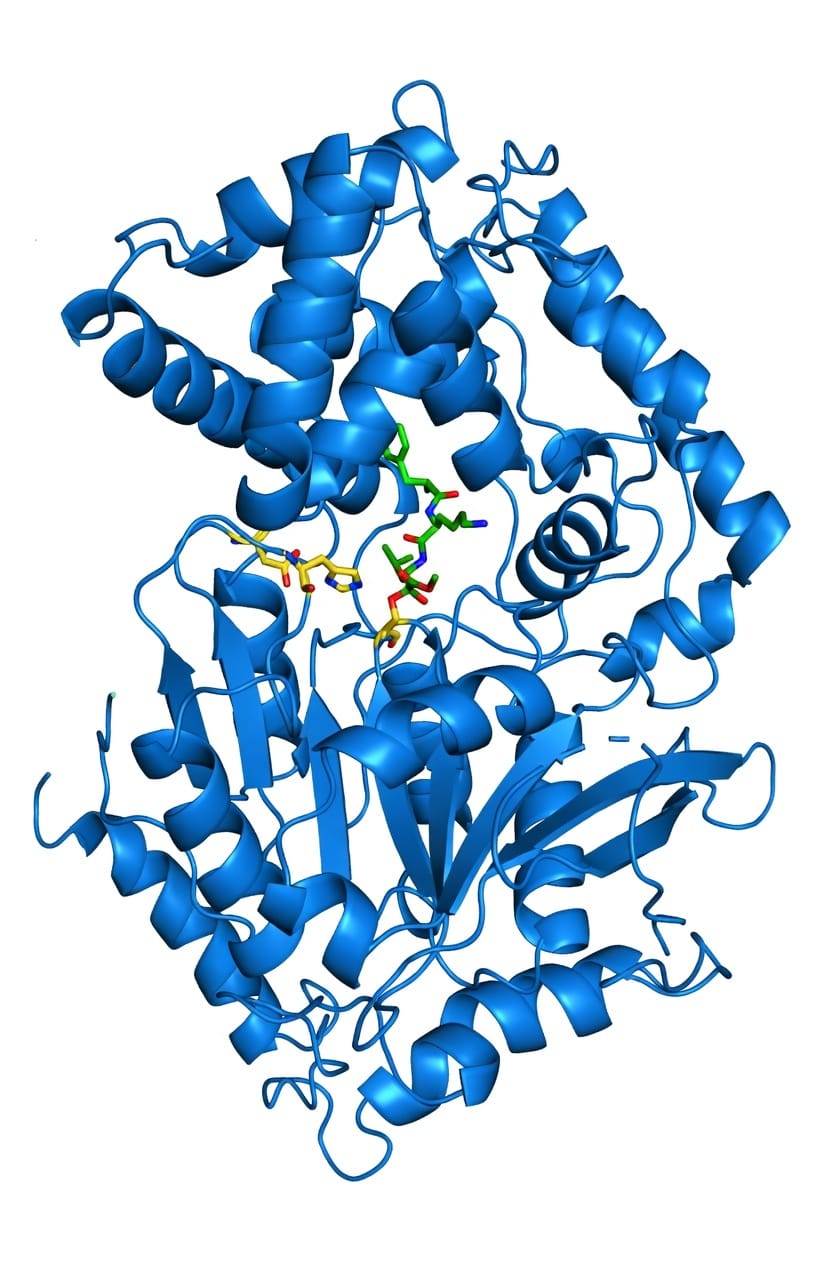
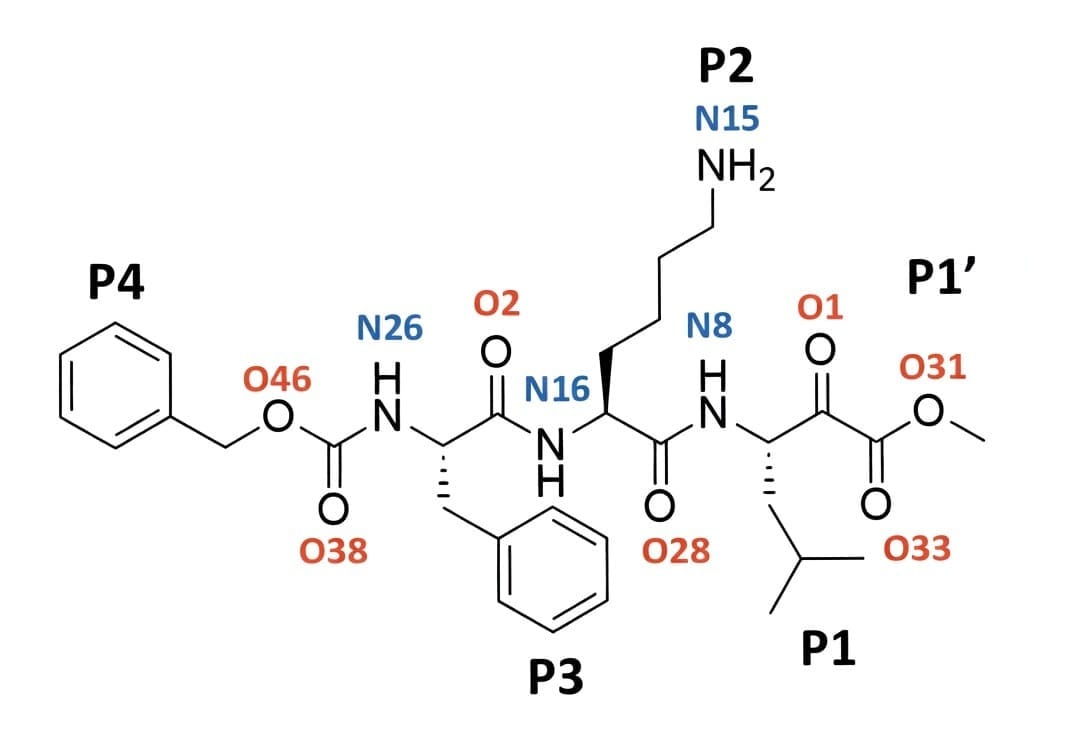
Using rational drug design and X-ray crystallography, the team developed two small molecules that bind tightly to the Hip1 enzyme, blocking its function. One compound in particular showed strong, sustained reductions in bacterial burden inside infected immune cells over time, while leaving the host cells unharmed.
The findings were published in the European Journal of Medicinal Chemistry Reports.

AI as a Research Partner
What makes this kind of work possible at a teaching university — rather than a large pharmaceutical company or major research institution — is, in part, the democratization of powerful computational tools.
In addition to X-ray crystallography, Goldfarb's lab has used AlphaFold, the AI system developed by DeepMind to predict the structures of other putative TB drug targets. Inhibition of secondary TB drug targets may increase the efficacy of current Hip1 inhibitors.
AlphaFold was central to the 2024 Nobel Prize in Chemistry. Combined with molecular docking software, it allows researchers to simulate how candidate compounds might interact with biological targets before a single molecule is synthesized in the lab.
"If you have the structure, you can see at the atomic level how the inhibitor binds," Goldfarb explained. "Without that, you're working blind."
It's an approach pharmaceutical companies are increasingly folding into their own pipelines, from early target identification all the way through optimization. For a lab without deep pockets, it's a force multiplier.
Not Just New Drugs — New Ways to Diagnose
Alongside the therapeutic research, Goldfarb's group is pursuing a second front: diagnostics, and the model he has in mind is one most people already know.
Think of a rapid COVID-19 test. You administer it at home, get a result quickly, and don't need a lab or a technician. Goldfarb wants to build something like that for tuberculosis.
Right now, no such thing exists. Detecting TB currently requires sophisticated and expensive instrumentation, as well as trained personnel, to analyze blood, urine and sputum (phlegm), not just saliva. That's a significant barrier in the regions where TB is most prevalent, and exactly the places least likely to have that infrastructure in place.
A rapid, easy test for TB is urgently needed in the global fight against the disease, which affects one-third of the world's population. Early detection is critical: the sooner a patient is identified and treated, the shorter the transmission chain, and the better the outcome.
If Goldfarb's team can develop a simple diagnostic tool, it could have a more immediate real-world impact than any new drug. Therapeutics take years to reach patients. A faster, cheaper test could change outcomes on the ground much sooner.
An Unlikely Collaboration
The research didn't happen in isolation. Goldfarb leads an international, multi-institutional team that includes researchers from Johns Hopkins University, the University of Adelaide, Utah State University, and California State University, Fresno.

UVU has also backed the work with institutional investment, supporting patent filings on the compounds and diagnostic assay. An original patent was filed a couple of years ago; a continuation was submitted more recently, a signal that the university sees something here worth protecting.
What the Lab Is Really Producing
There's another output from Goldfarb's lab that doesn't show up in journal articles: trained scientists.
Over the years, students have logged an estimated seven and a half cumulative years of work on this project: undergraduates learning molecular biology, data analysis, computational modeling, and how to explain their science to an audience. For students heading into the biotech workforce, that kind of sustained, real-stakes research experience is rare at a teaching-focused institution.
"This is an excellent example of how undergraduate-focused institutions like UVU can contribute to globally relevant scientific discoveries," Goldfarb said.
Where Things Stand
The compounds still need to clear significant hurdles. These include optimization, animal models, toxicity profiling, and, if everything goes right, multiple phases of clinical trials. The distance between a promising lab result and an approved therapy is vast, and most candidates don't make it.
Goldfarb knows this. He isn't overselling what he has.
What he does have is a target that behaves differently than most, a structural map of how his compounds bind to it, a published study, a global network of collaborators, and a lab full of students who've spent years learning to do serious science.
In a disease that has outlasted countless attempts to defeat it, that might be exactly where the next useful idea begins.
For detailed information about Goldfarb's research, check out "Discovery of highly potent α-keto ester-based peptidomimetic inhibitors of the Hip1 protease for the treatment of Mycobacterium tuberculosis" published in European Journal of Medicinal Chemistry Reports, 2025, volume 15.


